Author Information1
Ashok, K.2; Gottumukkala, A.3; Panesar, M.4; Song, J.5; Yang, R.6.
(Editor: Koizumi, N.7)
1 All authors are listed in alphabetical order
2 Westview High School, OR, 3 Thomas Jefferson High School for Science and Technology, VA,
4 Westfield High School, VA, 5 University of British Columbia, Canada, 6 Winston Churchill High School, MD, 7 George Mason University, VA
Background: The main obstacle to success is transplant rejection and managing the side effects of immunosuppressive medications. Induction and maintenance types of immunosuppressive medications are used in transplants. Induction therapy is used temporarily, only immediately following transplantation while maintenance immunosuppressive drugs are started and given long-term. Maintenance therapy can involve the use of antibodies or higher doses of medications. The standard triple medication regimen usually consists of the combination of a calcineurin inhibitor, an antiproliferative agent and a corticosteroid. These have been found to be more effective in balancing the risks of suppressed immune system with the benefit of organ transplant. However, the immunological risks (driven by age, preconditions, and other factors) of the child/pediatric patient should be considered, and the immunosuppressive therapy should be followed by therapeutic drug monitoring. [1]
A study conducted by Feng, Ekong & Lobritto (2012) reported that immunosuppression came with many toxic effects. Some of these include renal dysfunction, infection, metabolic perturbation, and malignancy [2]. To add on, Blondet, Healy & Hsu (2017) reported that the 2 most common immunosuppressants, tacrolimus and cyclosporine, were both known to have long-term side effects in children, which included nephrotoxicity, neurotoxicity, infection, and malignancy. Patient survival rates for pediatric liver transplant recipients after the use of immunosuppressive agents were 94% after one year, 91% after five years, and 88% after 10 years [3]. They also found that the side effects of immunosuppressive medications were severe in pediatric liver transplant recipients. Some immunosuppressive agents, such as tacrolimus, antithymocyte globulin, basiliximab, and others, might cause side effects including sensitivity, nausea, vomiting, diarrhea, high blood pressure, acne, abdominal pain, discomfort, mood swings and more. While immunosuppressants help with preventing rejection, it may be a challenge for pediatric patients since their reactions to the medications may be critical.
Objective: The study investigated how transplant outcomes (graft failure and patient mortality) vary by induction immunosuppressive therapy among pediatric liver transplant recipients.
Methods: A retrospective analysis was performed using the UNOS database between January 1, 2010 and June 30, 2022. The basic patient, donor and transplant characteristics were compared by basic demographics (age, sex and ethnicity) as well as induction and maintenance immunosuppressive therapies using T- and Chi-sq. tests. Survival curves and the estimates for transplant outcomes by induction and maintenance therapies were obtained using the Kaplan-Meier (KM) Product Limit method. In the analysis, patient death and graft failure were the end-points. Patients who did not experience any of the end-points or whose life and graft status was unknown were censored on the last follow-up or the last day of the study. Log-rank tests were used to test the equality of the curves. All analyses were done in R and the statistical significance was defined by p<0.05.
Results: There was a statistically significant difference in age between the group with graft failure (56.3) and the group without graft failure (54.5) (p < 0.001). Male patients were also more likely to experience graft failure. African American patients were more likely to experience graft failure, while Asian patients were less likely to experience graft failure (p < 0.001).
For the induction therapy, pediatric patients receiving Alemtuzumab (Campath) were much less likely to experience graft failure compared to other (antithymocyte globulin, basiliximab, and daclizumab) induction drugs (Figure 1: Panel A, p<0.001). Patient mortality survivals were similar, demonstrating that Alemtuzumab (Campath) reduces the patient mortality rate (Figure 1: Panel B, p<0.001).
Figure 1. Kaplan Meier Survival Curves by Induction Therapies

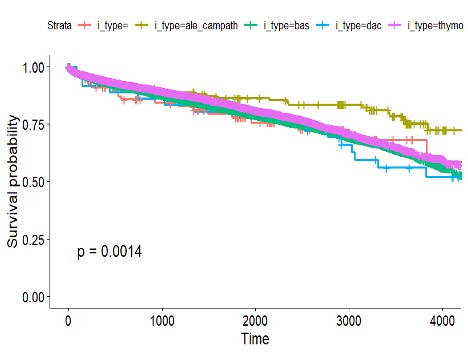
Panel A: Graft Survival Curves
Panel B: Patient Survival Curves
Figure 2. Kaplan Meier Survival Curves by Maintenance Therapy
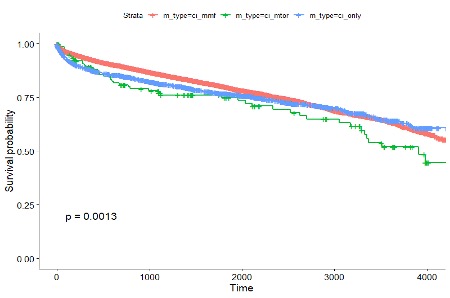

Panel A: Graft Survival Curves
Panel B: Patient Survival Curves
Figure 3. Kaplan Meier Survival Curves by Steroid Use
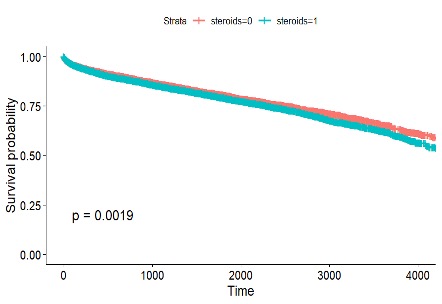
Panel A: Graft Survival Curves
Panel B: Patient Survival Curves
Conclusions: Pediatric liver transplant recipients who received alemtuzumab as the induction therapy had the best transplant outcomes while those recipients who received calcineurin inhibitors (tacrolimus or cyclosporine) with mammalian target of rapamycin (mTOR) as the maintenance therapy experienced the worst outcomes. Steroid use also worsened the outcomes.
References
- Lockridge, J. and Olyaei, A., 2019. Kidney Transplant-Immunosuppression and Rejection. Kidney Transplant Management: A Guide to Evaluation and Comorbidities, pp.29-42. https://link.springer.com/chapter/10.1007/978-3-030-00132-2_4
- Feng, S., Ekong, U.D., Lobritto, S.J., Demetris, A.J., Roberts, J.P., Rosenthal, P., Alonso, E.M., Philogene, M.C., Ikle, D., Poole, K.M. and Bridges, N.D., 2012. Complete immunosuppression withdrawal and subsequent allograft function among pediatric recipients of parental living donor liver transplants. Jama, 307(3), pp.283-293.
- Blondet, N.M., Healey, P.J. and Hsu, E., 2017, August. Immunosuppression in the pediatric transplant recipient. In Seminars in pediatric surgery (Vol. 26, No. 4, pp. 193-198). WB Saunders.
